Neuroinflammation in Epileptogenesis
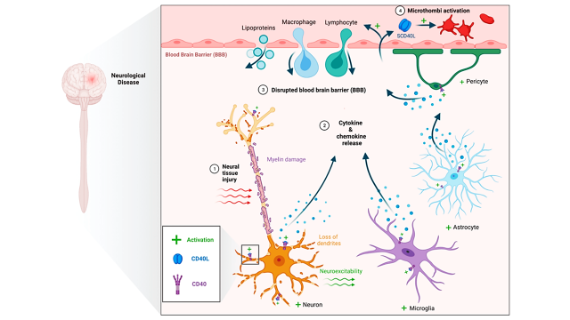
There is an urgent, unmet medical need for patients suffering from Temporal Lobe Epilepsy (TLE). This project aims to understand the underlying pathology and developmental mechanisms involved in developing TLE or limbic epileptogenesis (LE). We are investigating the role of the CD40 ligand (CD40L) in TLE.
CD40L activates molecular signaling through its CD40 receptor, leading to immune-inflammatory processes. We are using in vitro and in vivo models of epilepsy, genetic and molecular regulation of CD40L-CD40 system, behavior, and electrophysiology approaches, including histological techniques.
Our work will establish that the CD40L-CD40 interaction contributes to LE and will point toward avenues for developing immunotherapeutic treatments for TLE.
Glioblastoma multiforme and seizures
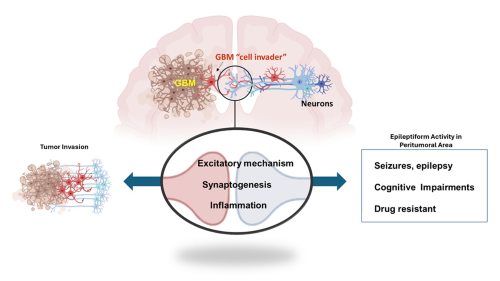
Glioblastoma multiforme (GBM) is a maligning brain tumor that has a poor survival rate. We focus on the physiopathology of GBM invasion of the brain. Our preliminary observations show that GBM increases the expression of Tumor Necrosis Factor Family receptors and spontaneous epileptiform activity.
This suggests that the GBM-neuron interaction could meditate tumor infiltration and proliferation. We are using behavior and electrophysiology approaches in the in vivo translational model of GBM, including an in vitro glioma/neuron co-culture following molecular and immunohistochemistry techniques. We will pave the way to discovering an early diagnosis and effective treatments for GBM, which are unmet medical needs.
Covid-19 and neural damage
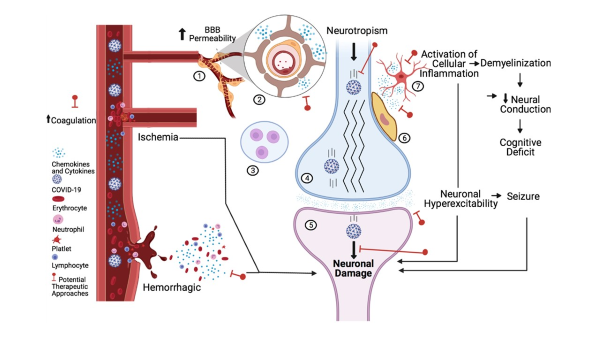
SARS-CoV-2, the etiologic agent of the 2020 Pandemic, can several dangerous neurological complications, suelicitch as acute ischemic or hemorrhagic stroke, etc.; these neurological complications are accompanied by central and peripheral cytokine storm.
This project aims to understand the unregulated inflammatory cascade triggered by the SARS-CoV-2- spike protein in the brain and peripheral system. We use clinical and in vivo models for SARS-CoV-2 to evaluate inflammation using mesoscale cytokine multiplex assays, electrophysiology, and immunohistochemistry. The identification of medication that can be used in neuroinflammation induced by SARS-CoV-2 is an unmet need in our society.
Planaria model for acute seizures
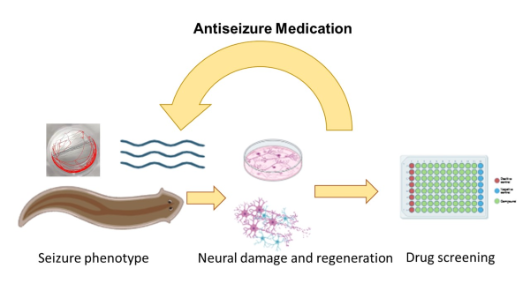
Experimental seizure models are very valuable for understanding the mechanisms of epilepsy and testing potential therapies. Planarians serve as cost-efficient and ethically favorable models for experimental seizures.
Unlike other invertebrates or zebrafish, planarians regenerate neurons and exhibit different behavior phenotypes, enhancing their value for antiseizure medication testing. We investigate seizure-induced neural regenerative mechanisms, neuronal connectivity changes, broader neurotoxicology research, genetic preservation, and modification after seizures.
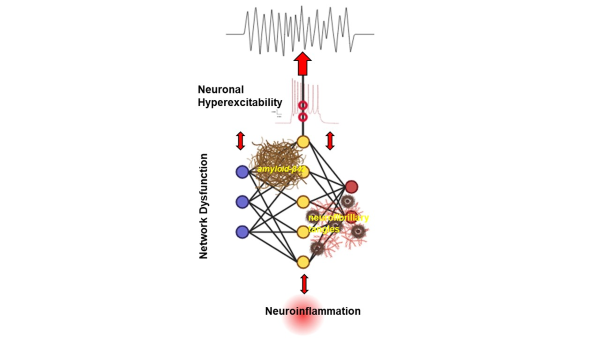
Alzheimer's Disease and neural network dysfunction
Alzheimer's disease (AD) devastates neuronal network functions. We plan to define the complex network of inflammation in the transgenic mouse models of AD, which are models that rapidly develop severe amyloid pathology.
Our studies articulate state-of-the-art methods in histology, electrophysiology, in vivo imaging, and mitochondrial functional analysis. Identifying the critical steps in the neuronal circuitry dysfunction involved in the early phase of AD could contribute toward finding new molecular targets to attenuate AD progression.
Methods and tools
Our experimental models include:
- Microelectrodes to study EEG, local field potential, and spike units in vivo
- Following neurostaining techniques to understand the cellular and subcellular structure of neurons
- Molecular biology to determine example inflammatory molecules using a multiarray system
- Cell culture
- Invertebrate systems to evaluate experimental compounds
- Signal analysis to evaluate progression of brain dysfunction in vivo